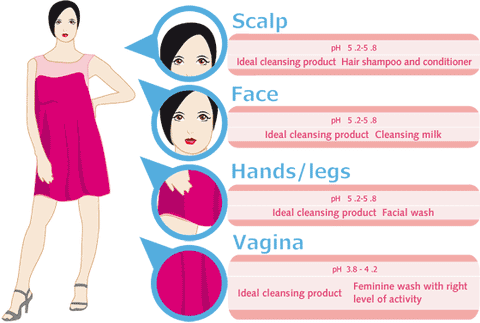
Just, what effects does pH have on the skin and how can it help your skin this winter?
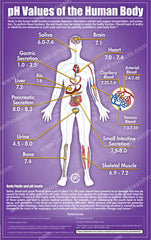
First as mot of us are not cosmetic chemist or dermatologist, you should know that pH is the measure of the skin acidity, it is normally pH 5.5. Inside your body the pH varies from 6 to 7.5 which is close to neutral (pH7).
When the pH is off, the skin does not operate at its optimal self and many key components of the skin are not synthesized:
- Key skin lipids processing enzymes are only active at an acidic pH. If the pH is off, lipids such as free fatty acids, phospholipids and ceramides will not be synthesized. These lipids are important components of the hydolipidic film as well as the cement binding the skin cells together. In conclusion the skin barrier that you really count on in the winter is damaged.
- Neutral to alkaline pH activates the enzymes responsible for desquamation. In other terms if the pH goes up the skin will start peeling, becomes thinner and less protective.
- Last the defensive, antimicrobial function of the skin is pH dependent. Pathogens such as P. Acnes bacteria, staphylococci, Candida and streptococci proliferate at neutral pH (pH7). On the other hand skin normal flora grows better at an acidic pH and if the pH stays up the bad bacteria start to grow.
For your skin to be at its best and happiest self, you need to make sure you create an optimum environment for your.
Here are few important tips to keep your skin at its best this winter:
- Switch your cleanser to a cleansing milk, drop the soap and wash with a shower gel. Soaps not only pull out the skin oils resulting in dehydration, but they also bring the skin's pH up high (pH 11 to 9) impairing the synthesis of skin binding lipids.

- Use a toner after you cleanse and after you rinse your skin with water in the morning (tap water is pH7). Toners will immediately bring your skin pH to 5.5 and wetting the skin before applying a product will enhance the penetration of the cream active ingredients.